Disease modeling and drug screening using human airway organoids: a systematic review
Article information
Abstract
Increasing levels of fine environmental dust particles due to industrialization and emerging respiratory illnesses, such as coronavirus disease 2019, pose serious threats to human life. The use of organoids for disease modeling and drug screening has been proposed as a new treatment approach for respiratory diseases. As discussed in this review, various pathogen models, genetic disease models, and patient-derived lung cancer organoid models have been reported for disease modeling and drug testing using human airway organoids. Despite these promising recent advances, several issues must be addressed before the disease modeling potential of human airway organoids can be fully realized. If systematic methods to produce mature airway organoids can be developed, and reproducible organoid models can be implemented using standardized protocols, airway organoids will likely become valuable respiratory disease models and drug screening tools.
Introduction
Respiratory diseases such as asthma, chronic obstructive pulmonary disease, acute respiratory infections, tuberculosis, and lung cancer represent a large and increasing public health burden; they are associated with high morbidity and mortality, and effective treatment regimens are currently lacking [1]. Furthermore, increasing levels of fine environmental dust particles due to industrialization and emerging respiratory illnesses, such as coronavirus disease 2019 (COVID-19), pose serious threats to human life.
Organoids have received attention recently as tools for studying embryogenesis, as well as for their potential applications as disease models and transplantable mini-organs. Recent advances have facilitated the development of methods to induce cellular differentiation and generate mature respiratory organoids; these methods have provided valuable insights into the developmental mechanisms of the upper and lower airways [2-5]. Moreover, researchers have attempted to transplant airway organoids into animal models [6]. However, implantation models in which the organoid lumen is in direct contact with the air are difficult to generate, and clinical trials for implants developed using embryonic system cells or induced pluripotent system cells are currently limited. Therefore, recent studies have focused on using airway organoids for modeling diseases and for high-throughput drug screening [4,7-10]. In particular, during the COVID-19 pandemic, human airway models were quickly used to study the pathology of infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), and drug testing was conducted to explore treatments for viral infections [11-18].
For organoids to become standard clinical in vivo mimetic models, they should ideally be derived from human cell sources. Therefore, in this review, we assessed the literature related to disease modeling and drug screening using human airway organoids, and discussed the future development and potential directions of airway organoid research.
Ethics statement: This study was a literature review of previously published studies and was therefore exempt from institutional review board approval.
Literature search method and selection
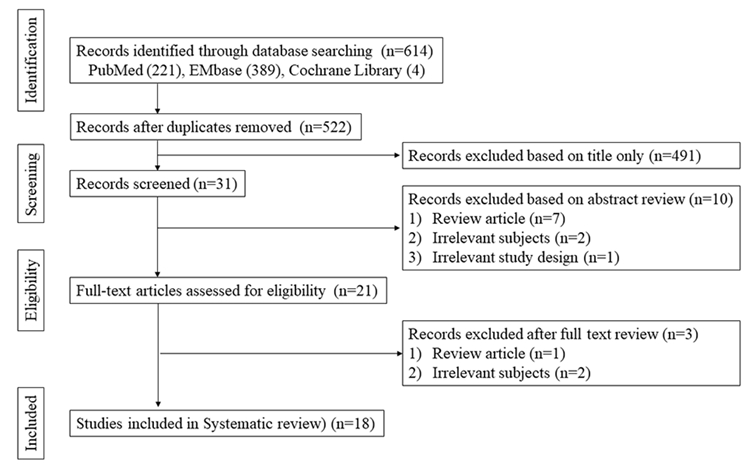
We retrieved research articles from the PubMed, Cochrane Central Register of Controlled Trials and Embase databases. The search period ranged from the inception of each database until March 2021. The search terms were as follows: “organoids,” “organ culture techniques,” “organogenesis,” “respiratory system,” “nose,” “trachea,” “bronchi,” “lung," “disease modeling,” “preclinical drug evaluation,” and “high-throughput.” Only studies published in English were reviewed. The reference lists of included articles were examined to ensure that no relevant studies were omitted. A detailed list of the search terms and queries is presented in Table S1. The inclusion criteria for this review were as follows: human organoid research, respiratory (airway) system research, and disease modeling or drug screening studies. We excluded case reports, review articles, and articles not written in English. All abstracts and titles of candidate studies were reviewed by two independent reviewers. A search flow diagram is presented in Fig. 1.
Disease modeling and drug screening research using human airway organoids
A summary of recent studies [7,8,10,15,19-28] in which human airway organoids were employed for disease modeling and drug testing is presented in Table 1 [7,8,10,12,15-28]. Since 2017, several reports of disease modeling and drug testing using human airway organoids have been published. Studies investigating the reactivity of organoids to influenza, parainfluenza, enterovirus, and various pathogenic bacteria have been conducted. Models of infections with viruses, Cryptosporidium parvum oocysts, and sporozoites were created by direct addition into organoid culture wells [7,8,10,20,22,26,27]. Furthermore, cystic fibrosis, a genetic disease that affects the respiratory system, and pneumonia, which also has a major genetic component, have been studied via human organoid models [8,20,23,25,28]. For the cystic fibrosis model, cystic fibrosis cell lines were obtained; they were made into patient-specific induced pluripotent stem cells (iPSCs) lines, and CFTR gene editing was performed to compare the results [8,25]. Congenital diaphragmatic hernia models were also prepared as lung organoids using iPSCs obtained from amniotic fluid and neonatal foreskin from children with congenital diaphragmatic hernia. In contrast, a model of Hermansky-Pudlak syndrome interstitial pneumonia was created with CRISPR-CAS9-induced deletion of the HPS1 gene [20]. An idiopathic pulmonary fibrosis model was created by treating fetal lung fibroblast organoids or PSC-derived mesenchymal organoids with exogenous transforming growth factor-β1 [28]. The use of patient-derived lung cancer organoids to screen for cancer drugs is another active area of research. In the lung cancer model, lung tissue was obtained from non-small-cell lung cancer patients and these tissues were produced as organoids. Through this model, the efficacy of various anti-cancer drugs was screened [19,24]. Recently, a human organoid infection model has been developed to screen and test candidate treatments for SARS-CoV-2. Youk et al. [18] built a 3-dimensional model to grow fluorescence activated cell sorting–classified human type 2 alveolar cells and airway epithelial cells into organisms, and then split them into pieces to enhance viral access to the apical cell surfaces. Tindle et al. [17] were able to established airway organoids with six epithelial cell types from the distal human lungs. These included type 1 and 2 alveolar cells, basal cells, club cells, goblet cells, and ciliated cells. Salahudeen et al. [16] developed chemically-defined culture protocols that could produce type 2 alveolar organoids and solid basal organoids with lumens. Han et al. [12,21] generated lung organoids that produced inflammatory reactions mimicking human COVID-19 infections, and tested high-throughput screening for US Food and Drug Administration-approved drugs and drugs known to inhibit SARS-CoV-2 entry. Mulay et al. [15] found that SARS-CoV-2 infection caused cell-autonomous and non-cell-autonomous cell death that could contribute to alveolar damage. They also found the virus targeted mainly ciliated cells in the airway. Candidates for the treatment of SARS-CoV-2 using airway organoids were imatinib, mycophenolic acid, quinacrine dihydrochloride, hydroxychloroquine, and remdesivir [15,21].
Limitations and future directions
Over recent years, methods for culturing airway organoids to investigate respiratory organogenesis have been well established [29,30]. Therefore, we focused on disease modeling and drug screening using airway organoids for this review, which demonstrated that multiple studies have been performed using organoids in various fields of respiratory research in recent years. For example, human organoids have been infected with bacteria and viruses in various studies, which confirmed their similarity to in vivo models; similar studies were conducted to test drug candidates. In addition, disease modeling using patient-derived cells to generate organoids has been performed and applied to the study of congenital and genetic diseases, as well as lung cancer. Organoid drug screening studies to develop effective treatments for SARS-CoV-2 are also in progress.
Despite these promising recent advances, several issues must be addressed before the disease modeling potential of human airway organoids can be fully realized. For example, the complete differentiation of human PSCs into specialized airway types is highly challenging, particularly for the differentiation of AEC1 cells and alveolar-like cell arrays [29]. To overcome this, researchers are exploring the possibility of using kidney capsules harvested from immunodeficient mice as niches to facilitate organoid maturation [9]. Another limitation of organoid models is their lack of reproducibility. This must be improved to establish organoids as reliable disease models, but doing so will require standardization of the organoid production process and the development of systematic high-throughput mass-screening tools. Accordingly, high-throughput processes are currently being developed to produce human airway organoids [31]. Moreover, the culture media used to induce organoids typically contain complex supplements, such as bovine extract or mouse sarcoma derivatives. Xenogenic-free, chemically-defined media are currently being developed [8,16,18,32] to further simplify and standardize the organoid production process.
Organoids are often referred to as “mini-organs”; however, it is currently challenging to produce large and complex structures with realistic organ sizes. When organoids reach a certain size threshold, the internal cells often undergo necrosis due to limitations in nutrient and oxygen exchange. To overcome this, bioengineered scaffolds are currently being developed [33]. During intestinal and respiratory organoid development, lumens form inside the spherical organoid structures. Physiological responses may differ between the respiratory system, which elicits immune responses to external substances detected by the epithelium, and the airway organoids, in which the apical lumen is situated within the spherical organoid structure. Techniques such as microinjection [26], cell shearing using Pasteur pipettes [7], enzymatic dissociation, organoid culture using a two-dimensional format [10] and apical-outward structure development using bioengineered niches [34] have been developed to mitigate these physiological differences.
Lastly, disease modeling and drug screening with organoids are currently limited to rare respiratory diseases with a relatively simple genetic basis. This has impeded organoid research using well-established models, such as asthma and chronic obstructive pulmonary disease, which are difficult to implement due to their complex etiology. In addition, efforts should be made to generate a cancer organoid model that is well integrated within a vascular structure to better reflect in vivo systems; current simple cancer organoid models comprise only cancer cells and surrounding fibroblasts.
Conclusion
The respiratory system, consisting of the upper and lower airways, is formed when the ectodermal and endodermal layers join together during embryogenesis; a key advantage of airway organoid models is that they include both developmental systems. If systematic methods to produce mature airway organoids can be developed, and reproducible organoid models can be implemented using standardized protocols, airway organoids will likely become valuable respiratory disease models and drug screening tools capable of replacing animal models for in vivo-mimicking in vitro studies.
Supplementary Information
Supplementary materials are presented online (available at https://doi.org/10.51335/organoid.2021.1.e8).
Additional file: Table S1
Search terms and queries.
Notes
Conflict of interest
Sung Won Kim has been an editor of Organoid since 2021. No other potential conflict of interest relevant to this article was reported.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2020R1I1A1A01051844) and the Bio & Medical Technology Development Program of the NRF funded by the Ministry of Science & ICT (2019M3A9H2032424, 2019M3E5D5064110). The sponsors had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional contributions
We thank Na Jin Kim (Medical Library, The Catholic University of Korea, Seoul, Republic of Korea) for help developing the search terms/queries and searching databases.
Data availability
Please contact the corresponding author for data availability.