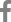|
 |
| Organoid > Volume 2; 2022 > Article |
|
Abstract
New drug development is currently very expensive and time-consuming. In addition, some drugs that are approved after animal and clinical trials have their approval revoked because of adverse effects. About 30% of such drugs have heart side effects. Conventional cell-based drug toxicity tests are performed under conditions entirely different from the in vivo environment, and animal testing for drug evaluation has limitations because of differences between species. Therefore, researchers are increasingly focusing on developing models that can overcome these limitations to enable accurate drug toxicity tests. This review outlines biomimetic in vitro heart platforms, such as heart organoids, 3-dimensional bioprinting, and heart-on-a-chip devices, and describes their advantages, limitations, future perspectives. The development and use of effective cardiac biomimetic models could contribute to the development of alternatives to animal testing by providing more specific information on drug metabolism and reducing the rate of failure in later stages of drug development.
Drug development is time-consuming, intensive, and costly, but few developed drugs are approved. The costs and time for achieving regulatory approval for a single drug are greater than $2.55 billion and between 10 and 15 years, respectively [1-3]. The high failure rate of drug development hinders investment in new drug discovery because it makes it challenging to generate profits [4]. To address this issue, many animal or cell-based experiments are typically conducted in the early stages of drug development.
Animal-based experiments require a considerable investment of financial resources and time, and also cause ethical debate. Furthermore, there are interspecies differences between humans and other animals. In cell-based experiments, cells are usually cultured under 2-dimensional (2D) conditions. These 2D cell culture models have advantages in terms of ease of handling and high throughput. However, they have limitations in drug toxicity and efficacy prediction because cells lose their inherent functionality during 2D culture, resulting in incorrect evaluation results [5,6]. For this reason, there is a need for 3-dimensional (3D) culture systems that reflect the actual organŌĆÖs structural, physiological, and environmental characteristics [7].
Microfluidic technology allows the fabrication of systems populated by human cells that recapitulate some aspects of organ function [8-10] and can be used to explore the origins of acute or chronic disease by probing critical molecular or cellular events. Organ-on-a-chip systems (OOCs) are biomimetic systems that can mimic the microenvironment of human organs, and are based on microfluidic cell culture devices with living cells in microchambers connected by a continuous perfusion system. Thus, OOCs are an effective alternative to animal testing that make it possible to overcome its limitations (cost, time, ethical issues, etc.) and can be useful as a platform in pre-clinical new drug development [6,11]. OOCs have been developed for several organs, including the lung [8], heart [12], eye [13], artery [14], and kidney [15]. OOCs enable studies of organ reactions to drugs or other chemicals that cannot be reproduced with conventional 2D or 3D culture systems.
Organoids are 3D cell aggregates formed through self-regeneration and self-organization from adult stem cells, embryonic stem cells (ESCs), or induced pluripotent stem cells (iPSCs) [16]. Organoids closely imitate the complex structure and functionality of human organs, such as the lung, liver, or brain [17]. Organoids allow the efficient use of existing 2D culture-based biochemical and cell biological analysis techniques while overcoming the limitations of the 2D cell culture method. Currently, intestinal, liver, lung, pancreatic, kidney, and uterine organoids have been reported from embryonic or adult stem cells [18]. Organoids can faithfully reproduce the physiological function of the human body, and the construction of organ-like structures from patient tissue samples enables disease modeling based on the patientŌĆÖs genetic information and drug screening through repeated tests [19].
Here we review the biomimetic in vitro heart platforms developed to study myocardial function and pathology from a microenvironmental perspective and highlight examples of heart-on-a-chip devices used as testbeds for new drug development.
Heart disease is one of the leading causes of death among Americans [20], and vascular dysfunction is a fundamental cause of morbidity and mortality in many diseases [21]. It is necessary to develop a more efficient evaluation system to replace animal testing when modeling diseases for new drug development, and human stem cell-based organoids and heart-on-a-chip devices are appropriate alternatives [22]. Current cardiac organoid production relies on direct cardiac differentiation from embryos or the production of cardiac tissue aligned from myocardial cells differentiated from a monolayer of human induced pluripotent stem cells (hiPSCs). Cases of the successful combination of cardiac organoids with biocompatible supports designed to mimic organ complexity have been reported [23], including cell-hydrogel matrices [24], a biomaterial-based microchamber [25], and 3D-printed biomaterials [26]. Recently, Mills et al. [24] developed a 96-well device for the functional screening of human cardiac organoids (hCOs) derived from human pluripotent stem cells and optimized its parameters, such as the extracellular substrate, metabolic substrate, and growth factor conditions that improve the function and maturity of cardiac tissue. This high-throughput hCO platform can be used to study the mechanisms of cell cycle arrest. In 2019, Lee et al. [26] reported a Bio3D-derived collagen technology (freeform reversible embedding of suspended hydrogels). The authors successfully 3D-printed the components of the human heart, and the 3D-printed heart accurately reproduced the patient-specific anatomical structure determined by microcomputed tomography. Ideally, non-genetic and genetic human heart diseases could be modeled in vitro using cardiac organoids. In 2020, Richards et al. [27] made hCOs using a non-adhesive agarose hydrogel template. They stimulated the hCOs with the neurotransmitter noradrenaline after myocardial infarction and reconstructed the characteristics of myocardial infarction. In combination with genome editing technology, hCOs can be used to accurately introduce mutations and obtain an innovative and personalized treatment platform for disease modeling. The clustered regularly interspaced short palindromic repeat/Cas9 system is a genome editing technique that can be used to correct and mitigate disease-causing mutations [28]. Although they have not yet been used to create a fully functional organism, complex hCOs are expected to be used in a wide range of fields, from observing heart disease progress, optimizing drug design, and evaluating drug toxicity to providing tools for preclinical trials (Fig. 1) [23,27,29-31].
Studies using 3D heart-on-a-chip devices have mainly focused on myocardial modeling. The myocardium is the main constituent tissue of the heart. In the basal myocardium, cardiomyocytes are present at 2 to 5 cell thicknesses and are surrounded mainly by heart fibroblasts within an extracellular matrix (ECM) containing collagen type ŌģĀ [32]. Several research groups have created heart-on-a-chip devices by simulating these heart structures and environments. In 2016, Marsano et al. [33] manufactured biomimetic heart chips using micro-processed polydimethylsiloxane (PDMS). Zoning was performed using PDMS membranes on the chip, and cardiomyocytes were cultured on coating materials in the upper part of the divided space. Markers expressed by cultured myocardial cells were identified to confirm that cell culture was performed well, and when stimulation was applied, the degree of change in the number of beats was measured and cell proliferation was verified. This platform provided a standard functional 3D heart model in the field and demonstrated the possibility of creating an innovative and inexpensive screening platform to improve the predictive ability of in vitro models (Fig. 2A). Morimoto et al. [34] designed a method to make fibrous 3D cell structures derived from hiPSCs to quantify the contractile force of myocardial cells. Myocardial cells were cultured in a patterned hydrogel structure with a fixed frame, and hiPS-derived cardiomyocyte (hiPS-CM) fibers with myocardial cells were successfully produced. The shrinkage force of hiPS-CMs was determined accurately according to the direction of the fiber. In addition, isoproterenol and propranolol were administered to confirm the drug reactivity of hiPS-CM fibers by observing changes in contraction frequency and the force of myocardial fibers (Fig. 2B). Grosberg et al. [35] used PDMS to develop a thin elastic film with a surface texture and implanted muscle cells on the membrane to form engineered muscle membranes (Fig. 2C). As the cardiomyocytes contracted, the film curled to one side. By measuring the degree of curling, differences in cell contraction on the PDMS film could be analyzed. This experimental system was suitable for single-muscle membrane measurements and multi-plate tests. Zhang et al. [36] aligned 3D human heart tissue patches manufactured using a hexagonal columnar PDMS structure. The structural and functional characteristics of these 3D tissue patches provided the closest in vitro approximation of human heart tissue along with the maturation of human ESC-derived myocardial cells. Schneider et al. [37] developed a new integrated device that enables the parallel generation of heart microstructures based on hiPSCs in specially designed multi-chamber systems that provide a precisely controlled physiological environment. The centrifugal cell-injection system was used to create bubble-free tissues. The viability and function of myocardial tissue could be maintained for an extended time period, and the applicability of drug testing in the chip system was demonstrated (Fig. 2D).
Owing to their small sample size and compatibility with patient-derived cells, microfluidic devices enable disease research under physiologically appropriate conditions. The increased availability of patient-specific iPSC lines has enabled the creation of physiologically related heart-on-a-chip systems for disease modeling in vitro [38]. In 2014, Wang et al. [39] used heart-on-a-chip technology to characterize abnormalities in cardiomyocytes obtained from hiPSCs generated from somatic cells of patients with Barth syndrome. This disease is caused by a mutation in a gene that encodes tafazzin, which is located in mitochondria, and leads to dilated cardiomyopathy (DCM) diagnosed immediately after birth. Here, hiPSC-CMs were characterized in terms of their metabolic phenotype and mitochondrial activity and combined with a platform designed to plant seeds in a muscle thin film in the next step to create an OOC for determining contractility disease-specific myocardial cells. Ren et al. [40] designed microstructure chip patterns using AutoCAD (Autodesk, San Rafael, CA, USA) and then made SU-8 (Microchem, Newton, MA, USA) molds based on these patterns to create PDMS heart chips. The protein markers expressed in cardiomyocytes cultured inside the chips were stained and measured by fluorescence microscopy. Under hypoxic conditions, these cardiomyocytes were verified to be applicable to actual disease models. In addition, this heart-on-a-chip system could be used to verify chemicals that might potentially be used for treatments through continuous monitoring of cell responses to these chemicals.
Another area of application of hiPSC-based heart-on-a-chip platforms is mechanical investigations of DCM caused by titin cleavage mutations [41]. Hinson et al. [41] have shown that manipulating cardiac tissues by using hiPSCs is a powerful system for evaluating diseases associated with titin gene mutations. They demonstrated that titin mutations interfere with the association between sarcomerogenesis and adaptive remodeling, leading to DCM (Fig. 3A). According to Tzatzalos et al. [42], hiPSC-CMs might have infinite potential for health with the use of disease-specific CMs to evaluate the efficacy of drugs against DCM. In experiments related to drug development, it is essential to evaluate the effect of drugs on cardiovascular tissue because cardiotoxicity is often seen in drug toxicity assessments and causes suspension of clinical trials or withdrawal of drugs from the market. Drugs may be withdrawn from the market because of previously unobserved toxic effects in animals resulting from off-target toxicity, including cardiac toxicity [42,43]. To address these concerns and better evaluate drugs before clinical trials, a platform for accurate evaluation in the drug discovery process is needed. Heart-on-a-chip models can be used as a platform to evaluate more accurately the efficacy and toxicity of drugs under conditions similar to in vivo conditions than cell-level studies. Mathur et al. [44] developed heart-on-a-chip devices by injecting cardiomyocytes differentiated from hiPSCs into PDMS chips with endothelium-like barrier microstructures. They assessed drug toxicity by injecting several types of drugs into vascular channels to examine changes in the myocardial rhythm at least 5 days after loading differentiated cells onto the chip. Verapamil, isoproterenol, metoprolol, and E-4031 were tested, and the beating rate of cardiomyocytes at different concentrations of each drug was analyzed with motion tracking software. The authors found that the drug response (IC50/EC50) was more reproducible than in cell-level studies (Fig. 3B).
A new hybrid strategy based on 3D bioprinting technology was proposed to produce myocardial and vascular systems in 2016 by Zhang et al. [45]. Microfibril hydrogel scaffolds bioprinted in endothelial cells formed vascular networks, and hiPS-CMs were added to generate an aligned myocardium. This 3D-myocardium organ chip produced a screening platform for cardiovascular drug testing. Zhang et al. [46] introduced a heart-on-a-chip device for evaluating drug efficacy using high-speed impedance detection technology. In this device, several parameters of myocardial cell rhythm were used to reveal the effectiveness of the drug. The results showed that this chip could determine drug efficacy or heart toxicity.
Organ-on-a-chip products are being launched by 12 companies in the United States, 6 in Europe (Switzerland, The Netherlands, Germany, and the United Kingdom), and one in Singapore; they are developing and selling single organ-on-a-chip or multi-organ-on-a-chip devices for various organs and tissues, including lungs, liver, nerves, kidneys, and blood vessels [47]. Among the commercialized bio-tissue chip products, 2 products can be called heart-on-a-chip devices. The SynTox 3D toxicology model (Synvivo, Huntsville, AL, USA), which injects myocardial cells into a universal chip design for use, and the uBeat (BiomimX, Milano, Italy) platform, which can provide controlled physical stimulation to microtissues such as heart muscle tissue. There are also 3 types of platforms capable of growing 3D cardiomyocytesŌĆöEHTs from EHT Technologies (Hamburg, Germany), Biowire II inaction from TARA (New York, NY, USA), and MyrPlate-Uniform from Myriamed (G├Čttingen, Germany) ŌĆöwhich induce the arrangement of myocardial cells with 2 vertical columns (Fig. 4).
Cardiac organoids and heart-on-a-chip devices and have high potential value as platforms for disease modeling and new drug development, and as tools for translational research. Despite its potential, this technology still has challenges to overcome.
PDMS is mainly used as the material of the microfluidic chip structure because PDMS is flexible and durable, has biocompatible characteristics, and facilitates the straightforward analysis of cell conditions using optical methods [48]. Despite the above advantages, some studies have reported disadvantages of PDMS [49,50]. The main problem with PDMS in microfluidic chip studies is the absorption of small molecules due to porous structures, affecting the concentration of water-soluble factors in the media and thereby influencing cell behavior and function. The porous matrix of PDMS can be an obstacle in some applications, such as cellular analysis or drug discovery applications. Other polymer materials are used to manufacture microfluidic devices according to the final properties and applications to overcome these problems [51]. Polytetrafluoroethylene (PTFE) can be used for microfluidic devices due to its thermo-processability, chemical inertness, and good electrical stability [52]. In 2021, Yao et al. [53] showed that PTFE could be used to inhibit the absorption of small hydrophobic molecules in PDMS. Another problem is that the reproducibility of OOC manufacturing varies depending on the properties of materials used, hydrophilic and hydrophobic surface treatment, the ECM used, and the storage conditions of the manufactured chip. In addition, the physiological activity of cells in a biological tissue chip may depend on whether immortalized cells, primary cells, or hiPSCs are used, on the composition of the medium that can be used to co-culture 2 or more cell types, and on the in-chip culture period. Furthermore, different cell types constituting one organ require different cell culture media. The use of cell culture medium optimized for a specific cell line is an obstacle to multiple cell interactions. There are also problems to be overcome in organoid platforms. Organoid culture is still expensive, and it is difficult to mass-produce organoids in comparison with cell culture. The ECM or scaffold used for 3D culture is both expensive and difficult to handle, making it difficult to automate the culture [54]. In addition, the histological structures and functions currently observed in organoids are insufficient compared to actual organs [55], and organoids are limited in size because they are entirely dependent on culture media instead of feeding through blood vessels. In order to improve organoids (in terms of shape and function) to a level sufficient for transplantation, optimization and scale expansion of culture methods are required.
The heart-on-a-chip model, which combines microfluidics and cell culture models, is similar to human organ tissue, and its excellence has been demonstrated. Recent advances in OOC technology are creating an available platform containing several flexibly connected organs to evaluate and predict the systemic effect of compounds efficiently [56]. These multi-organs-on-chip (MOOC) platforms are suitable for studying drug-related effects and improving drug safety testing in preclinical stages. In some cases, the presence or absence of cardiac toxicity in drugs that have undergone liver metabolism may change [57]. Therefore, research is underway to create a liver-heart model that can model and predict off-target heart toxicity related to the hepatic metabolism of drugs. A MOOC platform used to study cardiac toxicity induced by drugs and their metabolites was recently developed by Oleaga et al. [58]. This possibility of connecting multiple organs suggests that we can eventually create models that can potentially replace or at least complement existing animal models. In addition, as a pre-clinical trial model, organoid culture-based platforms have many potential applications, including customized drug screening for each patient and the application of regenerative medicine linked to genetic correction technology. However, many aspects need to be improved for practical use. Although most biomimetic chips and organoids have simulated the function and response of organs, they have not reached a level that would allow their use to successfully replace biological research. There is a need to continue improving many of the limitations of the platforms mentioned above. For drugs selected through a drug-screening platform to be used in actual clinical trials, it is necessary to establish a standard trial method and to compare and verify the actual clinical results and platform-based outcomes. These scientific and engineering efforts will provide more detailed information on drug metabolism and increase the reliability of human toxicity and efficacy evaluation, eventually reducing the failure rate in drug development and drastically reducing the cost and duration required to develop a new drug. In addition, this technology will contribute to the achievement of customized medical diagnosis and treatment by understanding the occurrence and progression of disease in each patient.
NOTES
Fig.┬Ā1.
Examples of human cardiac organoids. (A) Three-dimensional (3D)-bioengineered cardiac organoids. (Ōģ░) Visual assessment of environmental toxin effects on liver organoids by live/dead assay. (Ōģ▒) Environmental toxin effects on cardiac organoid ATP activity. Modified from Forsythe et al. [29], according to the Creative Commons license. (B) Development of a 3D post-myocardial infarction organoid model. (Ōģ░) Bright-field images and diameters of 4organoids on day 0 (D0) and day 10 (D10). (Ōģ▒) Control and cardiac organoid infarction protocol timeline using low oxygen (O2) and norepinephrine. (Ōģ▓) NADH autofluorescence from live 2-photon imaging of each organoids on D10 and NADH index quantification of control and infarct organoids. Modified from Richards et al. [27], with permission from Springer Nature.
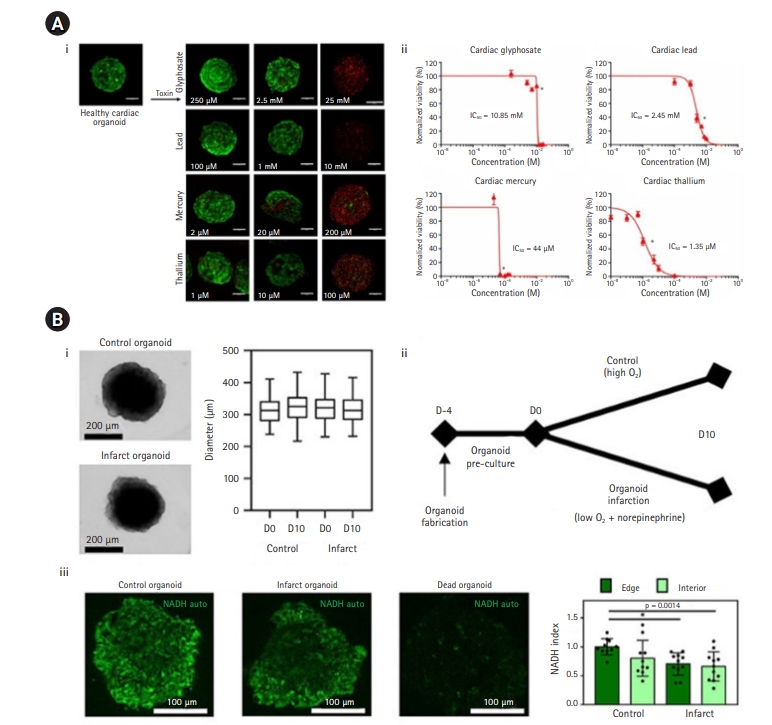
Fig.┬Ā2.
Summary of 3-dimensional (3D) heart-on-a-chip devices. (A) A biomimetic heart chip using micro-processed polydimethylsiloxane (PDMS). An individual PDMS microchamber was used. Cardiomyocytes were cultured in a coating material above the divided space. The lower end of the compartment was pressurized to deform the PDMS membrane and compress the 3D structure. Cell culture was performed well, as evidenced by measuring the degree of change in the contraction frequency of cardiomyocytes when stimulation was applied. Modified from Marsano et al. [33], with permission from Royal Society of Chemistry. (B) Method of making 3D biomimetic chips to quantify the contractile power of myocardial cells. Cardiomyocytes were cultured in patterned hydrogel structures. The drug reactivity of myocardial fibers was checked by observing changes in shrinkage frequency and force. Modified from Morimoto et al. [34], according to the Creative Commons license. (C) A muscle thin film (MTF) chip formed by engineering a muscle membrane. This MTF technique can be applied to various tests for smooth and striated muscles, including testing contractile properties. Modified from Grosberg et al. [35], with permission from Elsevier. (D) An integrated device that enables the creation of a heart microstructure in a specially designed multi-chamber system. The chip system could be used in various biomedical fields. Modified from Schneider et al. [37], according to the Creative Commons license. hiPS-CM, hiPS-derived cardiomyocyte.
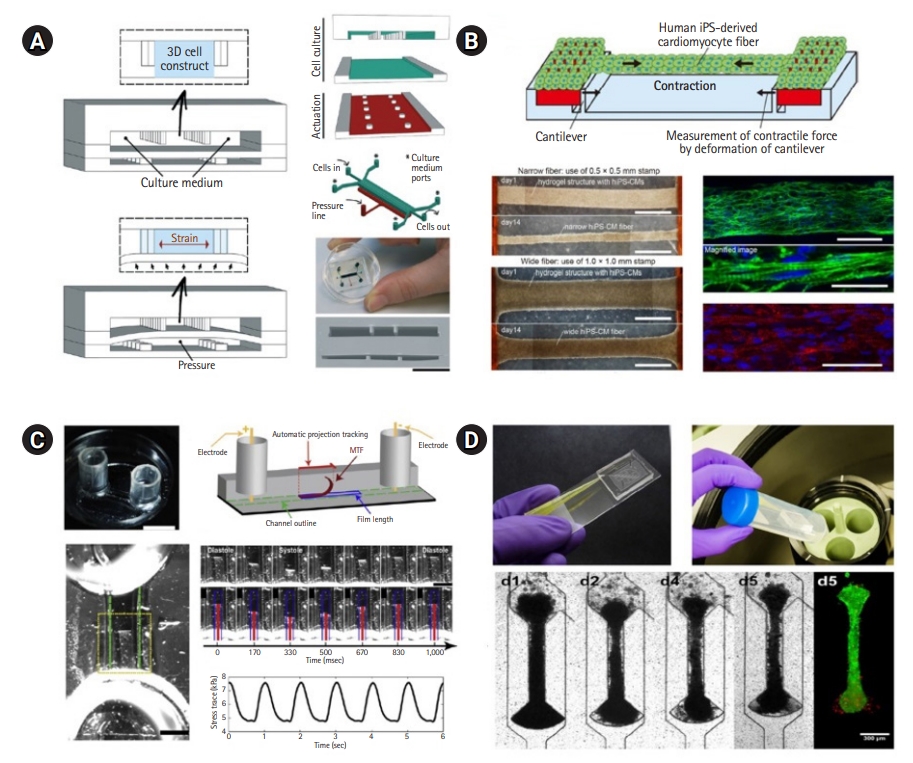
Fig.┬Ā3.
Representative examples of heart-on-a-chip devices used in disease modeling and drug efficacy evaluation. (A) A study of incomplete penetrance of dilated cardiomyopathy (DCM) caused by a titin cleavage mutation using a heart-on-a-chip platform. This study demonstrated that the titin mutation interfered with the association between sarcomerogenesis and adaptive remodeling, resulting in DCM, and revealed that cardiac tissue manipulated in human induced pluripotent stem cells could be used as a robust evaluation system associated with titin mutation. Modified from Hinson et al. [41], with permission from AAAS. (B) A schematic diagram of polydimethylsiloxane heart-on-a-chip devices with endothelium-like barrier microstructures. Drug toxicity was assessed by injecting several types of drugs into the vascular channel to examine changes in myocardial rhythm. The rate of the beating of myocardial cells at each drug concentration was analyzed by motion tracking software, and it was found that the drug response (IC50/EC50) was more reproducible than in cell-level studies. Modified from Mathur et al. [44], according to the Creative Commons license. ETPC, estimated therapeutic plasma concentration in patients.
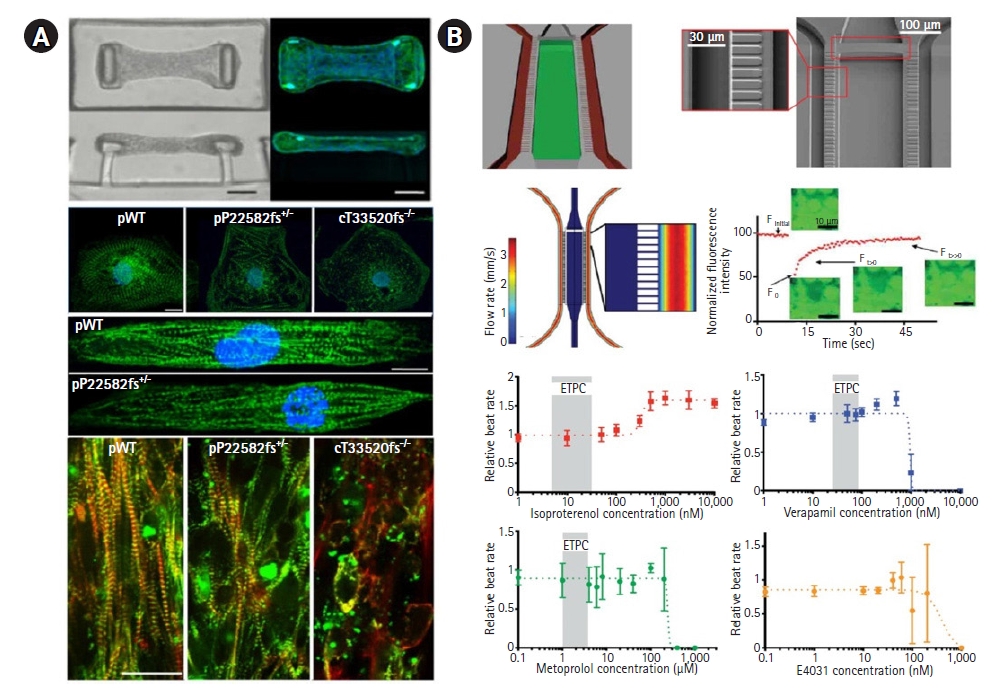
Fig.┬Ā4.
Summary of commercially available heart-on-a-chip products. (A) SynTox 3D toxicology model (Synvivo) containing the following components; (1) vascular channel with 2 inlet ports (A and E) connected to 2 outlet ports (B and F); (2) tissue chamber with 2 inlet ports (C) connected to 2 outlet ports (D). A general chip design is used to inject cardiomyocytes. (B) uBeat (BiomimX) platform can specifically stimulate the physical beat of the myocardium. (C-E) Biological tissue platforms capable of cultivating 3D myocardial tissue. EHTs (EHT Technologies) induce the arrangement of myocardial cells in 2 vertical pillars. In addition, Biowire II inaction (TARA) and MyrPlate-Uniform (Myriamed) are available.

References
1. DiMasi JA, Grabowski HG, Hansen RW. Innovation in the pharmaceutical industry: new estimates of R&D costs. J Health Econ 2016;47:20-33.


2. DiMasi JA, Hansen RW, Grabowski HG. The price of innovation: new estimates of drug development costs. J Health Econ 2003;22:151-85.


3. Dickson M, Gagnon JP. Key factors in the rising cost of new drug discovery and development. Nat Rev Drug Discov 2004;3:417-29.


4. Holmes A, Bonner F, Jones D. Assessing drug safety in human tissues: what are the barriers? Nat Rev Drug Discov 2015;14:585-7.


5. Sung JH, Esch MB, Prot JM, Long CJ, Smith A, Hickman JJ, et al. Microfabricated mammalian organ systems and their integration into models of whole animals and humans. Lab Chip 2013;13:1201-12.



6. van der Meer AD, van den Berg A. Organs-on-chips: breaking the in vitro impasse. Integr Biol (Camb) 2012;4:461-70.


7. Antoni D, Burckel H, Josset E, Noel G. Three-dimensional cell culture: a breakthrough in vivo. Int J Mol Sci 2015;16:5517-27.



8. Huh D, Matthews BD, Mammoto A, Montoya-Zavala M, Hsin HY, Ingber DE. Reconstituting organ-level lung functions on a chip. Science 2010;328:1662-8.



10. Low LA, Mummery C, Berridge BR, Austin CP, Tagle DA. Organs-on-chips: into the next decade. Nat Rev Drug Discov 2021;20:345-61.



12. Grosberg A, Alford PW, McCain ML, Parker KK. Ensembles of engineered cardiac tissues for physiological and pharmacological study: heart on a chip. Lab Chip 2011;11:4165-73.



13. Chan YK, Sy KH, Wong CY, Man PK, Wong D, Shum HC. In vitro modeling of emulsification of silicone oil as intraocular tamponade using microengineered eye-on-a-chip. Invest Ophthalmol Vis Sci 2015;56:3314-9.


14. Yasotharan S, Pinto S, Sled JG, Bolz SS, G├╝nther A. Artery-on-a-chip platform for automated, multimodal assessment of cerebral blood vessel structure and function. Lab Chip 2015;15:2660-9.


15. Wilmer MJ, Ng CP, Lanz HL, Vulto P, Suter-Dick L, Masereeuw R. Kidney-on-a-chip technology for drug-induced nephrotoxicity screening. Trends Biotechnol 2016;34:156-70.


16. Willyard C. The boom in mini stomachs, brains, breasts, kidneys and more. Nature 2015;523:520-2.



18. Li M, Izpisua Belmonte JC. Organoids: preclinical models of human disease. N Engl J Med 2019;380:569-79.


20. Heron M, Anderson RN. Changes in the leading cause of death: recent patterns in heart disease and cancer mortality. NCHS Data Brief 2016;254:1-8.
21. Rajendran P, Rengarajan T, Thangavel J, Nishigaki Y, Sakthisekaran D, Sethi G, et al. The vascular endothelium and human diseases. Int J Biol Sci 2013;9:1057-69.



22. Zwi-Dantsis L, Gepstein L. Induced pluripotent stem cells for cardiac repair. Cell Mol Life Sci 2012;69:3285-99.



23. Zhao D, Lei W, Hu S. Cardiac organoid: a promising perspective of preclinical model. Stem Cell Res Ther 2021;12:272.




24. Mills RJ, Titmarsh DM, Koenig X, Parker BL, Ryall JG, Quaife-Ryan GA, et al. Functional screening in human cardiac organoids reveals a metabolic mechanism for cardiomyocyte cell cycle arrest. Proc Natl Acad Sci U S A 2017;114:E8372-81.



25. Hoang P, Wang J, Conklin BR, Healy KE, Ma Z. Generation of spatial-patterned early-developing cardiac organoids using human pluripotent stem cells. Nat Protoc 2018;13:723-37.




26. Lee A, Hudson AR, Shiwarski DJ, Tashman JW, Hinton TJ, Yerneni S, et al. 3D bioprinting of collagen to rebuild components of the human heart. Science 2019;365:482-7.


27. Richards DJ, Li Y, Kerr CM, Yao J, Beeson GC, Coyle RC, et al. Human cardiac organoids for the modelling of myocardial infarction and drug cardiotoxicity. Nat Biomed Eng 2020;4:446-62.




28. Driehuis E, Clevers H. CRISPR/Cas 9 genome editing and its applications in organoids. Am J Physiol Gastrointest Liver Physiol 2017;312:G257-65.


29. Forsythe SD, Devarasetty M, Shupe T, Bishop C, Atala A, Soker S, et al. Environmental toxin screening using human-derived 3D bioengineered liver and cardiac organoids. Front Public Health 2018;6:103.



30. Li RA, Keung W, Cashman TJ, Backeris PC, Johnson BV, Bardot ES, et al. Bioengineering an electro-mechanically functional miniature ventricular heart chamber from human pluripotent stem cells. Biomaterials 2018;163:116-27.



31. Goldfracht I, Protze S, Shiti A, Setter N, Gruber A, Shaheen N, et al. Generating ring-shaped engineered heart tissues from ventricular and atrial human pluripotent stem cell-derived cardiomyocytes. Nat Commun 2020;11:75.




33. Marsano A, Conficconi C, Lemme M, Occhetta P, Gaudiello E, Votta E, et al. Beating heart on a chip: a novel microfluidic platform to generate functional 3D cardiac microtissues. Lab Chip 2016;16:599-610.


34. Morimoto Y, Mori S, Sakai F, Takeuchi S. Human induced pluripotent stem cell-derived fiber-shaped cardiac tissue on a chip. Lab Chip 2016;16:2295-301.


35. Grosberg A, Nesmith AP, Goss JA, Brigham MD, McCain ML, Parker KK. Muscle on a chip: in vitro contractility assays for smooth and striated muscle. J Pharmacol Toxicol Methods 2012;65:126-35.



36. Zhang D, Shadrin IY, Lam J, Xian HQ, Snodgrass HR, Bursac N. Tissue-engineered cardiac patch for advanced functional maturation of human ESC-derived cardiomyocytes. Biomaterials 2013;34:5813-20.



37. Schneider O, Zeifang L, Fuchs S, Sailer C, Loskill P. User-friendly and parallelized generation of human induced pluripotent stem cell-derived microtissues in a centrifugal heart-on-a-chip. Tissue Eng Part A 2019;25:786-98.



38. Doherty EL, Aw WY, Hickey AJ, Polacheck WJ. Microfluidic and organ-on-a-chip approaches to investigate cellular and microenvironmental contributions to cardiovascular function and pathology. Front Bioeng Biotechnol 2021;9:624435.



39. Wang G, McCain ML, Yang L, He A, Pasqualini FS, Agarwal A, et al. Modeling the mitochondrial cardiomyopathy of Barth syndrome with induced pluripotent stem cell and heart-on-chip technologies. Nat Med 2014;20:616-23.




40. Ren L, Liu W, Wang Y, Wang JC, Tu Q, Xu J, et al. Investigation of hypoxia-induced myocardial injury dynamics in a tissue interface mimicking microfluidic device. Anal Chem 2013;85:235-44.


41. Hinson JT, Chopra A, Nafissi N, Polacheck WJ, Benson CC, Swist S, et al. Heart disease: titin mutations in iPS cells define sarcomere insufficiency as a cause of dilated cardiomyopathy. Science 2015;349:982-6.



42. Tzatzalos E, Abilez OJ, Shukla P, Wu JC. Engineered heart tissues and induced pluripotent stem cells: macro- and microstructures for disease modeling, drug screening, and translational studies. Adv Drug Deliv Rev 2016;96:234-44.


43. Ferri N, Siegl P, Corsini A, Herrmann J, Lerman A, Benghozi R. Drug attrition during pre-clinical and clinical development: understanding and managing drug-induced cardiotoxicity. Pharmacol Ther 2013;138:470-84.


44. Mathur A, Loskill P, Shao K, Huebsch N, Hong S, Marcus SG, et al. Human iPSC-based cardiac microphysiological system for drug screening applications. Sci Rep 2015;5:8883.




45. Zhang YS, Arneri A, Bersini S, Shin SR, Zhu K, Goli-Malekabadi Z, et al. Bioprinting 3D microfibrous scaffolds for engineering endothelialized myocardium and heart-on-a-chip. Biomaterials 2016;110:45-59.



46. Zhang X, Wang T, Wang P, Hu N. High-throughput assessment of drug cardiac safety using a high-speed impedance detection technology-based heart-on-a-chip. Micromachines (Basel) 2016;7:122.



48. Nielsen JB, Hanson RL, Almughamsi HM, Pang C, Fish TR, Woolley AT. Microfluidics: innovations in materials and their fabrication and functionalization. Anal Chem 2020;92:150-68.


49. Toepke MW, Beebe DJ. PDMS absorption of small molecules and consequences in microfluidic applications. Lab Chip 2006;6:1484-6.


50. Ren K, Zhou J, Wu H. Materials for microfluidic chip fabrication. Acc Chem Res 2013;46:2396-406.


51. Niculescu AG, Chircov C, Birca AC, Grumezescu AM. Fabrication and applications of microfluidic devices: a review. Int J Mol Sci 2021;22:2011.



52. Liao S, He Y, Chu Y, Liao H, Wang Y. Solvent-resistant and fully recyclable perfluoropolyether-based elastomer for microfluidic chip fabrication. J Mater Chem A 2019;7:16249-56.

53. Yao J, Guan Y, Park Y, Choi YE, Kim HS, Park J. Optimization of PTFE coating on PDMS surfaces for inhibition of hydrophobic molecule absorption for increased optical detection sensitivity. Sensors (Basel) 2021;21:1754.



54. Langhans SA. Three-dimensional in vitro cell culture models in drug discovery and drug repositioning. Front Pharmacol 2018;9:6.



- TOOLS
-
METRICS

-
- 0 Crossref
- 0 Scopus
- 6,166 View
- 125 Download
- ORCID iDs
-
Sungwoo Cho

https://orcid.org/0000-0003-4617-3209Sungho Ko

https://orcid.org/0000-0002-8108-8918 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print