Strategies for vascularization in kidney organoids
Article information
Abstract
The establishment of protocols for differentiating kidney organoids from human pluripotent stem cells (hPSCs) has potential for the application of kidney organoids in regenerative medicine. However, the primary obstacle to the regenerative application of hPSC-derived kidney organoids is precise vascularization due to the lack of vasculature in hPSC-derived kidney organoids. In this article, we review the recent methodologies for developing vasculature of kidney organoids to overcome this limitation of kidney organoids, together with a discussion of their clinical applications.
Introduction
Chronic kidney disease (CKD), which affects 5% to 7% of the adult population worldwide, leads to end-stage renal disease (ESRD) and results in substantial morbidity and mortality [1–6]. ESRD can be treated by renal replacement therapy, including dialysis and renal transplantation. However, both renal replacement therapies are limited—the mortality and morbidity rates in patients on dialysis are much higher than those in the general population, while transplantation is limited by the shortage of donor organs and the need for lifelong immunosuppressive therapy. Because the global prevalence and annual incidence of CKD are increasing annually, new therapeutic options are urgently needed.
Organ regeneration has recently emerged as a new therapeutic strategy, which has potential for medical therapeutic benefits. Kidney organoids can be generated from human pluripotent stem cells (hPSCs) in vitro, and may be an attractive option for regenerative medicine for CKD [7–10]. However, the primary obstacle to the regenerative application of hPSC-derived kidney organoids is precise vascularization due to the lack of vasculature in hPSC-derived kidney organoids. In this article, we review the recent methodologies for developing vasculature in kidney organoids to overcome the limitations of kidney organoids, together with a discussion of their clinical applications.
Ethics statement: This study was a literature review of previously published studies and was therefore exempt from institutional review board approval.
Vasculature of the kidney
The human kidneys are highly vascularized organs that normally receive approximately 20% of the cardiac output even though they make up less than 1% of the total body mass [11]. The kidney vasculature has a unique and complex architecture that is essential for kidney function, including glomerular filtration, regulation of blood pressure, and maintenance of acid–base and electrolyte balance [12–14].
The human kidney is supplied with blood through the renal artery, which branches directly from the abdominal aorta. The human renal arteries branch into anterior and posterior branches at the site of the renal hilum and then into segmental arteries [11,15]. The segmental arteries branch into interlobar arteries, which form the arcuate arteries around the border between the cortex and medulla. Each arcuate artery divides into multiple interlobular arteries that give rise to the afferent arterioles, which connect with individual glomeruli [11,15].
After entering the glomerulus, the afferent arteriole branches to form the glomerular capillary network, in which endothelial cells constitute the glomerular filtration barrier with the glomerular basement membrane and podocytes form the glomerular filtration barrier. The glomerular capillaries drain into efferent arterioles. The highly fenestrated endothelial cells in this network are part of the glomerular filtration barrier and contribute to its development and maintenance through paracrine cross-talk with podocytes and mesangial cells.
Overall, the kidney vasculature is extremely complex, making it one of the greatest challenges in organ regeneration technologies.
Generation of hPSCs‑derived kidney organoids
Based on an understanding of embryogenesis, several groups have developed methods for differentiating kidney organoids from hPSCs [8,16–19]. Developing these protocols for the generation of kidney organoids in vitro is based on providing appropriate growth factors and WNT signals with CHIR treatment at the right time. hPSC-derived kidney organoids contain nephron progenitor cells, which could give rise to podocytes, cells of the Bowman’s capsule, and tubule epithelial cells, with appropriate morphological patterning, protein staining, gene expression, and histological features [8,16–19]. Comparisons between kidney organoids and human fetal kidneys have shown that these organoids mimic the natural embryonic development of human fetal kidneys [18,20]. Kidney organoids derived from hPSCs have potential applications in regenerative medicine, as well as in modeling renal diseases, drug screening, and nephrotoxicity testing of compounds [17,21–31].
Differences in protocols among study groups have resulted in the production of nephron-like structures of different quality and with different ratios of cell types [20]. Wu et al. [20] compared 2 differentiation protocols—the Morizane protocol and the Takasato protocol—by single-cell transcriptomics of fetal and adult kidney cells. They showed that both protocols generated a diverse range of kidney cells, with differing ratios; the organoid-derived cell types were immature, and 10% to 20% of cells were non-renal. The proportion of endothelial cells was very small, whereas the human kidney consists of a large number of endothelial cells. Standardized and advanced protocols are needed for the clinical application of hPSC-derived kidney organoids.
Strategies for the vascularization of hPSC-derived kidney organoids
As described above, one of the major functions of glomeruli in the kidney is to filter blood to generate urine. Therefore, an adequate vasculature is essential for kidney function. However, the differentiation of vascular endothelial cells in kidney organoids is limited, and most glomeruli within organoids remain avascular in vitro. Researchers have made many efforts to enhance the vascularization of kidney organoids, and some challenges are discussed in Fig. 1.
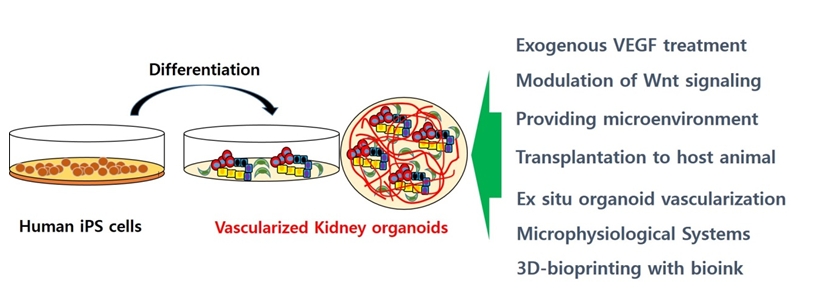
Strategies of vascularization in kidney organoids. iPSC, induced pluripotent stem cell; VEGF, vascular endothelial growth factor; 3D, 3-dimensional.
1. Exogenous vascular endothelial growth factor treatment
Czerniecki et al. [32] increased the vascularization of kidney organoids through the addition of exogenous vascular endothelial growth factor (VEGF) during the differentiation process. In kidney organoids with exogenous VEGF treatment, the melanoma cell adhesion molecule protein was specifically expressed in CD31+ endothelial cells, occupying large portions of the surface area, consistent with the identification of these cells as endothelial cell progenitors. Endothelial cell-specific growth receptors were detected at low levels by single-cell RNA sequencing, despite substantial expression of their ligands in neighboring cells. The researchers postulated that while VEGF treatment greatly increases the number of endothelial cell progenitors in organoid cultures, only a small minority of these cells reach a mature endothelial cell differentiation state similar to that found in vivo.
2. Modulation of WNT signaling
During development, accurate patterning of different nephron segments along the proximal-distal axis is crucial for kidney function later in life [33]. Multiple signaling pathways are implicated in patterning the proximal-distal axis of the nephron, such as WNT, BMP, and Notch [33]. Low et al. [33] enhanced the formation of a vascular network by precise modulation of WNT signaling. They postulated that the modulation of WNT signaling in nascent renal vesicles could alter the relative ratio of proximal versus distal segments within kidney organoids. Through precise modulation of WNT signaling, they reported that VEGFA was highly expressed by podocytes and that differentiating podocytes may autonomously secrete VEGFA to support the resident vasculature without exogenous VEGFA treatment.
3. Providing a microenvironment by biomaterials
Chick chorioallantoic membrane (CAM) is a naturally highly vascularized extraembryonic tissue that has been used in tumor angiogenesis research and for the grafting of biomaterial. It provides a soft in vivo microenvironment that promotes organoid vascularization. Garreta et al. [34] showed that transplantation of kidney organoids onto CAM enhanced vascularization in ovo and promoted the maturation of nephrons with endothelial invasion into glomeruli in transplanted kidney organoids. CAM-implanted kidney organoids had morphological features that reflected their functional differentiation compared with in vitro conditions.
Garreta et al. [34] also showed that the stiffness of the CAM-microenvironment was recapitulated in vitro by fabricating compliant hydrogels. CAM-like soft substrates accelerated the formation of more nephron structures than those produced on rigid substrates. While kidney organoids cultured in rigid conditions formed vascular endothelial cells surrounding the nephron structures, but lacked a vascular network, blood vessels from the CAM invaded the implanted kidney organoids, which interacted with the glomerular structures, and chick blood was able to circulate through the organoids. These findings suggest that the microenvironment can modulate the differentiation of hPSC-derived kidney organoids.
4. In situ kidney organoid vascularization by transplantation to a host animal
Some recent reports have described in detail the vascularization of human kidney organoid grafts beneath the kidney capsule and suggested increased maturity of these structures [15,35–38]. Taguchi et al. [8] reported engraftment after the transplantation of metanephric nephron progenitors differentiated from mouse embryonic stem cells by their own protocol into the subcapsular space of immunodeficient mice. At 1 week after transplantation, massive tubulogenesis was observed in the transplanted grafts. Podocyte-like cells in the graft expressed vascular growth factors. They also showed that the transplanted glomeruli vascularized with the host circulation and contained red blood cells. Sharmin et al. [36] reported that the glomeruli in transplanted spheres with human induced pluripotent stem cell (iPSC)-derived nephron progenitors were vascularized with host mouse endothelial cells with further maturation, although capillary loops rarely formed. They differentiated glomeruli-like structures from human iPSCs expressing green fluorescent protein at the NPHS1 locus, which were transplanted into the mouse kidney subcapsular space using spacers that released the tension of host kidney capsules and treated with VEGF. On day 10 after transplantation, immature glomerular formation accompanied by blood vessels was observed. On day 20 after transplantation, the transplanted grafts were enlarged and the transplanted glomeruli were vascularized with the host mouse endothelial cells. Human iPSC-derived podocytes had numerous cell processes accumulated around the fenestrated endothelial cells, forming slit diaphragm-like structures, which were a more mature feature than found in kidney organoids in vitro. van den Berg et al. [38] also showed that transplanted kidney organoids derived from hPSCs had a matured glomerular filtration barrier and tubular epithelium with host mouse-derived vascularization. They transplanted kidney organoids derived from hPSCs into mouse kidney subcapsular spaces. On days 7 and 14 after transplantation, they reported that functional glomerular perfusion with connections to host mouse vascular networks was observed by in vivo imaging. On day 28 after transplantation, more mature features (i.e., slit diaphragm-like structures and more polarized and segmental specialized tubular epithelium) were observed than on day 7 after transplantation, indicating that kidney organoids matured progressively with the time of transplantation.
We previously transplanted kidney organoids derived from iPSCs into mouse kidneys [39]. We also observed some vascularization of podocytes and maturation in tubules in our grafts, similar to previous studies [39]. We observed human endothelial cells derived from the transplanted kidney organoids, but the proportion of endothelial cells in the graft was small. Mouse endothelial cells were abundantly observed in the transplanted kidney organoid grafts, as well as within the glomerulus-like structures. These findings indicate that endothelial cells from the host mouse kidney had extensively infiltrated into the transplanted kidney organoids and formed a vascular network.
It remains unclear why the vascularization of kidney organoids in vitro is limited, but organoids are readily vascularized after transplantation in vivo. It seems that mechanical flow from the host vasculature might play an important role.
Despite the vascularization of kidney organoids, immaturity-related concerns remain. Podocytes do not appear to form fully mature foot processes, and instead are decorated by apical microvilli, which are distinct structures [39]. The lack of fully mature features, such as aquaporin-1 expression, compared to neighboring mouse kidney tissue also gives rise to concerns related to immaturity concerns despite the vascularization after transplantation [39].
5. Ex situ kidney organoid vascularization
Based on the evidence of in situ vascularization following transplantation, ex situ transplantation might contribute to the vascularization of kidney organoids [7,40–42]. The host body can be used as a bioreactor in a secondary location prior to re-transplantation at the target site in a second operation. Previous studies have suggested that the lymph node functions as an effective bioreactor for many types of engrafted cells [42]. The lymph node is able to provide endothelial capillaries and, therefore, critical nutrients and growth factors from the blood to transplanted organoids. Furthermore, lymph nodes contain fibroblastic reticular cells and other stromal cells that secrete chemokines, which enhance cell recruitment, growth, expansion, and survival [40,41]. Thus, lymph nodes may also be an attractive target for potential kidney tissue regeneration.
One previous study implanted human kidney progenitors from human fetal kidneys into mouse lymph nodes [7]. The implanted kidney progenitors differentiated into organoids and subsequently matured in the lymph node’s in vivo environment. Those implanted in vivo organoids exhibited excretory, homeostatic, and endocrine functions, with a significant amount of host-derived vasculature.
6. Microphysiological systems
Blood flow is essential for adequate kidney vascularization based on a previous study showing that chemically induced heartbeat arrest in zebrafish impaired the integration of the vasculature into glomeruli [43]. Microphysiological systems can be used to form functional vascular networks. Homan et al. [44] induced substantial vascularization and morphological maturation in kidney organoids in vitro using flow on millifluidic chips. Their method showed that vascularized kidney organoids cultured under flow, especially high flow, had more mature podocytes and tubular compartments, with enhanced cellular polarity and adult gene expression, than did static controls [44]. Interestingly, the endogenous upregulation of VEGF under high fluidic shear stress allows vessels to reach glomerulus-like compartments in time to invade, rather than wrap Bowman’s capsule–like structures within these kidney organoids in vitro, indicating the development of capillary loop stage-like glomeruli.
We developed a kidney organoid-on-a-chip system providing fluidic flow mimicking shear stress with optimized extracellular matrix conditions [45]. We showed that the kidney organoids cultured in our microfluidic system showed more matured podocytes and vascular structures as compared to static culture conditions. Additionally, the kidney organoids cultured in microfluidic systems showed higher sensitivity to nephrotoxic drugs as compared with those cultured in static conditions. We also demonstrated that physiological flow played an important role in maintaining a number of physiological functions of kidney organoids. Therefore, our kidney organoid-on-a-chip system could provide an organoid culture platform for in vitro vascularization in the formation of functional 3-dimensional (3D) tissues.
Flow-enhanced vascularization and maturation of kidney organoids on 3D-printed microfluidic chips has emerged as the most promising method of in vitro organoid vascularization, which can be applicable for investigating organogenesis, nephrotoxicity, tubular and glomerular disease, and kidney regeneration with a simple millifluidic chip.
Future directions and conclusions
We have summarized the strategies for the vascularization of hPSC-derived kidney organoids (Fig. 1). Vascularization of kidney organoids with an elaborate network of arteries, veins, and capillaries is one of the major challenges for their clinical application. An issue for the vascularization of kidney organoids is the need to generate larger arteries, both inside and outside of the kidney organoids, that will form an accurate vascular network similar to that of an in vivo kidney as well as a “kidney on a chip.” 3D bioprinting techniques with kidney-specific bioinks, such as kidney decellularized extracellular matrix, will provide a more precise and reproducible method for constructing kidney organoids or even whole organs [46–52].
Current protocols for the generation of kidney organoids with a real vascular network harmonizing angiogenesis and vasculogenesis remain to be established. Overcoming these hurdles will require a better understanding of vasculature development at the single-cell level at different developmental stages. A comprehensive knowledge of the molecular mechanisms of renal vascular development can then be applied to drive the development of more mature renal endothelial cells. These efforts, when integrated with emerging techniques such as 3D bioprinting and tissue engineering, will open new avenues toward the generation of “real” in vitro kidneys in the future.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, Science and Technology (NRF-2021R1A2B5B01001793, NRF-2019M3A9H2032546). This research was also supported by the Korean Fund for Regenerative Medicine funded by Ministry of Science and ICT, and the Ministry of Health and Welfare (21B0601L1-01).
Data availability
Please contact the corresponding author for data availability.
